CDX Models for Oncology Studies
Our cell line-derived xenograft (CDX) model studies can be offered as subcutaneous, disseminated, or orthotopic in both mice and rats, and can also be used in our humanized mouse models for evaluating immunotherapies within an intact human immune system.
Human tumor CDX models involve implantation of commercially available tumor cell lines or patient-derived xenograft cell lines into immunodeficient mice to conduct in vivo efficacy evaluation of potential cancer therapies, including targeted therapy, cytotoxic drugs, antibody therapies, and viral therapies.
Oncology Preclinical Study Planning Toolkit
Take control of your efficacy study by getting instant access to study pricing, cancer model data, and connection to our oncology specialists, so that you feel supported on your oncology efficacy journey.
Find the Right Model
Our broad range of tumor lines gives you a wide variety of testing alternatives for your agent. We evaluate our xenograft model portfolio on a regular basis for response to standards of care to aid the design of combination studies.
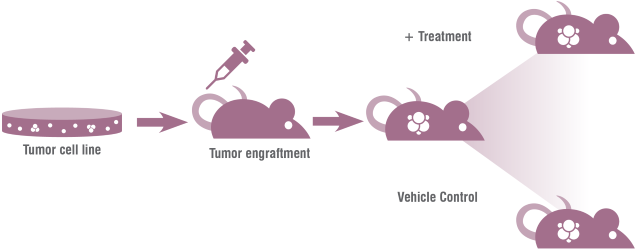
Cell Line-Derived Xenograft (CDX) Model
Figure 1: Diagram of Cell Line-Derived Xenograft, CDX Model
In addition to helping you select the most appropriate CDX model for your agent, our scientific staff can fully profile your agent in a variety of tumor types or in combination with the appropriate clinical agent.
Charles River offers a wide range of histotypes, including broad models like non-small cell lung cancer and breast or colon cancers, as well as tumor models with a high medical need, like ovarian cancer, leukemia, lymphoma, or prostate cancer.
Available CDX Cell Line Models by Indication
See the table below for our available cell line models. If you require a model that is not listed here, please contact us and we can investigate validating a model for you.
| Histotype | Available Cell Lines |
|---|---|
| Adrenal | H295R |
| Bladder | RT112*, SW780* |
| Brain | SF-268*, U251*, U87-MG*, Daoy (meduloblastoma) |
| Breast | BT474*, HCC-1806*, HCC-1937, HCC-1954*, HCC70*, JIMT-1*, MCF-7*, MDA-MB-231*, MDA-MB436*, MDA-MB-453, MDA-MB-468*, SK-BR-3, T47D*, CAL51 |
| Colon | 94*, CL-34*, COLO 205*, DiFi, DLD-1*, HCC-2998, HCT 116*, HCT-15*, HT-29*, KM12*, KM20L2, LoVo*, LS-174T*, LS411N*, RKO*, SW48*, SW480*, SW620*, SW403 |
| Endocervical | KB* |
| Endometrial | MFE-280*, SNG-M |
| Epidermoid Carcinoma | A-431* |
| Esophageal | OE19* |
| Ewing’s Sarcoma | RD-ES* |
| Gastric | 3011*, FU97, IM95*, MKN1, MKN45*, N87*, NUGC-4*, SCH*, SNU-5* |
| Head and Neck | CAL-27*, FaDu*, SNU-1076* |
| Leukemia | ARH-77*, CCRF-CEM*, EM-2*, HEL92.1.7*, HL-60*, JURKAT, JURL-MK1*, K562*, Kasumi-1*, KCL-22, KG-1*, KG-1a*, MEC-1*, MEG-01*, MOLM-13*, MOLT-4*, MV4-11*, NALM-33*, NALM-6*, NKM-1*, NOMO-1*, OCI-AML2*, OCI-AML3*, RS4;11*, SET2*, THP-1* |
| Liver | Hep G2*, Hep3B*, SK-HEP-1*, SNU-398*, SNU-449, SS49, SNU-739, SNU-761,SNU-878 |
| Lung | H1581, H226, H441 |
| Lung (Non-Small Cell) | 66*, 983*, A-427*, Calu-1, CALU-6*, H1155, H1299*, H1437*, H1568*, H1650*, H1666, H1703*, H1975*, H1993, H2122*, H23*, H292*, H460*, H520*, H522*, H647*, H727*, H810*, HCC-1171, HCC-44*, HCC827*, SK-MES-1* |
| Lung (Small Cell) | DMS 114*, DMS 273, H1963*, H211*, H526*, H69*, H727*, H82*, SHP-77* |
| Lymphoma | Daudi*, DoHH-2*, Granta 519*, HUT-78*, JEKO-1*, KARPAS-299*, Mino*, Namalwa*, OCI-LY7, Raji B*, Ramos*, REC-1*, RL*, SU-DHL-1*, SU-DHL-4*, U-937, WSU-DLCL2*, Z-138* |
| Melanoma | 520*, A2058*, A375*, CHL-1*, COLO 800*, HT-144, IGR-1, IGR-37*, LOX*, SK-MEL-28, UACC-62* |
| Mesothelioma | MSTO-211H* |
| Multiple Myeloma | H929*, IM-9*, KMS-11*, L-363*, LP-1*, MM.1R*, MM.1S, OPM-2*, RPMI 8226*, U-266* |
| Neuroblastoma | IMR-32, SK-N-AS*, SK-N-MC* |
| Osteosarcoma | 143B* |
| Ovarian | A2780*, AG6000, BG1, IGR0V1*, OVCAR-3*, OVCAR-4, OVCAR-5*, OVCAR-8*, OVSAHO, SK-OV-3*, TOV-21G* |
| Pancreas | AsPC-1*, BxPc-3*, Capan-1*, CAPAN-2*, HPAC*, HPAF II*, KP4*, MIA PaCa-2*, PANC1*, PSN-1* |
| Pleuramesothelioma | H-MESO-1 |
| Prostate | 22Rv.1*, DU145*, DuCap, LNCaP*, LNCaP-FGC*, PC3*, PC-3M*, Vcap* |
| Renal | 786-0*, Caki-1, G-401*, G-402* |
| Sarcoma | HT-1080*, SJSA-1* |
| Thyroid | 8505C*, FTC-238*, K1, CAL62 |
| Uterine | ECC-1, HEC-1-A, HEC-1-B |
| Uterus | 1138* |
Please note that all cell lines with an * are available with profile data. For those that have not yet been profiled for response to current standards of care, tumor growth curve data is available on request.
Efficacy evaluation in CDX models can be combined with:
- Hematology (CBC/Diff) analysis
- Pharmacokinetic or bioavailability sampling and analysis
- Histopathology or tissue sampling
- Biomarker screening, sampling, or evaluation
- In vivo microdialysis to determine pharmacological effects with high sensitivity and temporal resolution
- In vivo implantable microdevices; simultaneous in vivo testing of multiple drugs, drug doses, or drug combinations in a single tumor
Learn More About Our CDX Models
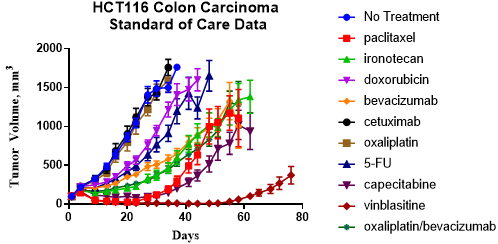
Figure 2: Charles River’s CDX models have extensive standard of care data for combination design
Cell Line-Derived Xenograft - Cancer Model Database
Support your in vitro, in vivo, and ex vivo studies with the following features:
- New user-friendly search and easy-to-navigate menus
- New model data, including HLA typing, growth curves, and tumor images
- New multi-parameter search options for all tumor model types (PDX and CDX)
If you’re looking to purchase oncology research models for use in your own efficacy studies, please visit our research models pages or see our xenograft data collection for help in selecting the best animal model.
Learn More About Our CDX Models
Frequently Asked Questions (FAQs) for Cell Line-Derived Xenograft (CDX) Mouse Models
-
What is a cell line-derived xenograft (CDX) model?
A cell line-derived xenograft or CDX model is a gold-standard model used for the research and testing of anti-cancer therapies. Human tumor samples are cultured as cell lines and implanted into mouse models to test the efficacy of anti-tumor compounds in vivo.
-
Why should I choose Charles River to run my cell line-derived xenograft (CDX) models?
In addition to over 200 CDX models, Charles River also has more than 80 proprietary PDX-derived cell line models across the full range of tumor histotypes, providing full translational relevance into the clinic.
- How can Charles River’s integrated oncology services help my study?